Lecture 1:
Introduction to Thermodynamics – Role of thermodynamic Principles in real-life situations – active learning and attitude of a true student – Course Outline – Syllabus
- System, Definition and Types of Systems – Closed, Open, Isolated; System properties
Lecture 2:
- Postulates of Thermodynamics:
- (n+2) rule
- Existence of stable equilibrium
- State
- Point and Path Functions
- Work
- Deriving expression for Work from first principles (force balance)
- Adiabatic work interactions (most direct path from one point to another)
- Postulate III of Thermodynamics: Change of state is possible in at least one route and the adiabatic work is identified from the corresponding end states
Lecture 3:
- Definition of Heat
- Postulate IV of Thermodynamics or Zeroth Law of Thermodynamics: A&B are in equilibrium; B&C are in equilibrium, then A&C are in equilibrium
- First law of Thermodynamics: dQ = dU + dW; importance of sign conventions
- First law as applied to closed systems
- Ideal gas concept – applied to gases; Enthalpy, Internal Energy and relation between the heat capacities of gases
Lecture 4:
- First law as applied to open systems
- Work refinements – Types of Work: Electrical, Mechanical, Surface Tension, Elastic, Kinetic Energy, Rotational Relativistic
- Additional Enthalpies
- Sensible Heat
- Latent Heat: -Change of Phase, Chemical Reaction, Nuclear Reaction
Lecture 5:
Flow Systems:
Force balance on steady-state, steady flow systems
- Isothermal Expansion
- Converging Nozzle
- Heat Exchanger
- Adiabatic periodic piston
- Ideal isothermal turbine
- Ideal adiabatic turbine
Lecture 7:
Work – Energy cycles with examples – Flow Systems – Batch
systems – Bernoulli equation as a special case of 1st law of
thermodynamics
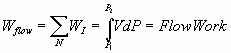 ;
;
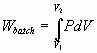
Ideal Isothermal Turbine
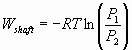
The difference in the work produced in an adiabatic batch
vs. flow system is related to the difference in D H
& D U = D (PV)
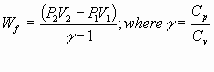
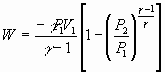
= -nCp (T2-T1)
Example 4: Adiabatic Expansion @ Constant Volume
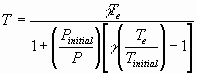
For P >> Pintial; T
à g Te
Lecture 8:
Definition of Path Function
Part II of Example 4 – Constant Adiabatic Expansion @
Constant Pressure
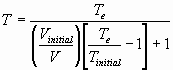
Example 8 – Home Work problem 2.32: Real Gas work:
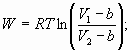 Isothermal batch
systems – Home Work Problem 2.29:
Isothermal batch
systems – Home Work Problem 2.29:
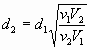 ; H2-H1
=
; H2-H1
=  (v12-v22)/2
(v12-v22)/2
Lecture 9:
Historical approach to the second law of thermodynamics –
Carnot (heat engine) and Clausius (entropy)
Examples demonstrating the Entropy phenomena, Concept of
Equilibrium, Heat and Work
Definition of Reversible process – Rankine cycle
Concept of Thermodynamic Temperature
Lecture 10:
Definition of Thermodynamic Efficiency (h
) and its derivation
Introduction to Entropy – Definition, Concept of Entropy
from heat engines
For any isolated system D Ssystem
³ 0;
For any system D Ssystem+D
Ssurroundings ³ 0
Homework Problems: 5.5, 5.8, 5.14
Entropy and 2nd law of Thermodynamics –
Definition and derivation of thermodynamic efficiency – Efficiency limits –
functionality
Carnot Cycle: Definition and derivation of Carnot
efficiency
Clausius’ Theorem: Any process can be broken into
equivalent adiabatic and isothermal steps
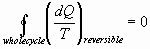
For a process with less than a complete cycle, entropy is defined as:
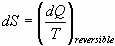
Entropy is a state function; Entropy of the Universe is never conserved
Lecture 11:
Combined 1st and 2nd law of Thermodynamics:
Conservation of Energy with respect to Entropy
Ideal gases and 2nd law of Thermodynamics: Isochoric process,
Isobaric Processes, Isothermal processes, Processes from (P1, V1,
T1) to (P2, V2, T2) – with respect
to Entropy and Adiabatic reversible processes: Isentropic processes
(n+2) rule for a single phase system with respect to Entropy
Examples of Second law of Thermodynamics, Reversible work, to calculate
change in Entropy: Bird problem, Hilsch tube etc.
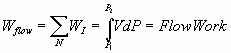 ;
;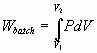
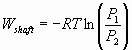
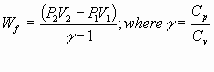
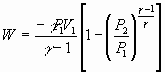

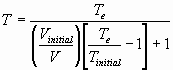
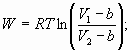 Isothermal batch
systems – Home Work Problem 2.29:
Isothermal batch
systems – Home Work Problem 2.29:
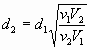 ; H2-H1
=
; H2-H1
=